The Neural Control of Adaptive Behavior
Survival requires responding to constantly changing conditions. Over time, evolution has selected for neural circuits that ensure robust and adaptive responses to survival needs. My lab is focused on uncovering the neural circuits and mechanisms that prioritize these behaviors.
One key survival need for all organisms is ensuring ‘adaptive food intake’1,2. In other words, it is necessary to obtain sufficient food, often in the face of competing needs, but also to avoid overconsumption. Our current studies explore the cellular, molecular, and network mechanisms that regulate hunger and seek to understand how hunger circuits influence other survival needs (Figure 1). By examining how the brain senses and responds to fundamental survival needs, we strive to define general principles of how the brain guides behavior in a dynamic world.
Objective 1: Determine how activity in hypothalamic circuits is regulated to influence food intake.
Adaptive food intake requires energy intake to match energy expenditure. For this energy balance to occur, physiological nutrient status must be determined by the brain, and neural activity in hunger circuits must rapidly respond to subsequent changes. Classical lesion studies dating back to the 1940s identified the hypothalamus as a key regulator of food intake and energy expenditure3. More recently, cell types in the hypothalamus that promote or inhibit food intake have enabled a more precise investigation of the neural control of food intake4-6.
In the arcuate nucleus of the hypothalamus, two neuron populations are critical regulators of food intake. Neurons expressing agouti-related protein (AgRP neurons) are active during hunger, whereas neurons expressing pro-opiomelanocortin (POMC neurons) are active during satiety. Through in vivo monitoring of these energy-sensing neuron populations, we found that the activity of AgRP and POMC neurons is rapidly regulated by calorie sensing in the gut7 (Figure 2A). Building on this observation, the lab is defining the gut-brain pathways that rapidly signal the presence of ingested nutrients. We have identified both vagal8 and spinal9 gut-brain pathways that rapidly alert the brain to the addition of nutrients in the gastrointestinal tract (Figure 2B).
To better understand how hunger circuits function in a dynamic world, we are exploring how energy expenditure (a homeostatic challenge) influences hunger circuit function. In nature, this may happen due to the need to evade predators or while seeking prey. In the laboratory, we are testing how dynamic changes in energy expenditure (i.e., exercise) modify neural activity in hunger circuits. We have demonstrated that exercise rapidly changes neural activity in hypothalamic circuits, including those that regulate hunger10,11 (Figure 2C). Our emerging studies indicate that exercise leads to plasticity and increased activity in other hypothalamic neurons that influence metabolism.
Having identified how food and exercise impact brain activity, our current work is investigating how this information is integrated in the brain to affect various aspects of feeding behavior (Figure 2D). In one project, we are seeking to understand the pathways through which nutritive information is transmitted to the hypothalamus. In a second project, our goal is to understand the cellular, molecular and neural circuit basis for how and why the brain rapidly responds to changes in metabolic need. We found that exercise leads to rapid changes in hunger circuits (Figure 2C, 2D), but what is the mechanistic basis of these changes? These studies couple classical lesion approaches with state-of-the-art neural activity monitoring to examine how physiological state and behavior influence neural activity in the awake, behaving animal.

Objective 2: Identify and characterize additional neural nodes in the network that regulates hunger.
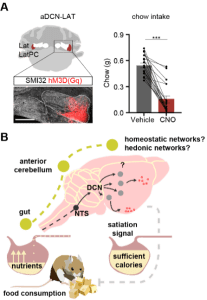
Over the last 15 years, neural circuits that regulate food intake have been identified and characterized. However, pharmacological agents targeting these known neural circuits have not led to successful weight loss therapeutics. It is therefore likely that our current knowledge is incomplete and that unknown yet essential nodes to the neural networks that regulate food have yet to be discovered. To obtain an unbiased assessment of the neural circuits that regulate food intake, and to identify new neural nodes that regulate feeding, we performed a collaborative study to image neural activity (fMRI BOLD) in human subjects with a genetic disorder (Prader Willi Syndrome) that causes constant hyperphagia, or a lack of being satiated following a meal. Through this effort, we identified a dramatic dysregulation in cerebellar output circuits. We characterized these cerebellar feeding circuits in rodent experiments and identified the precise neuron type and function of the cerebellum in feeding: our data show that activity in a small subset of glutamatergic neurons in the deep cerebellar nuclei dramatically reduces food intake by decreasing meal size (Figure 3A).
In our current work, we are exploring how these cerebellar neurons are integrated into the canonical networks known to control food intake. We hypothesize that cerebellar activity generates a forward model that predicts calorie content of a meal, enabling consumption of appropriate meal sizes. These experiments aim to understand: (1) if activity in the cerebellar output neurons predicts the calories consumed in a meal; (2) how activity in cerebellar output neurons influences homeostatic/hypothalamic or hedonic/midbrain circuits; and (3) how neural activity in the cerebellum is regulated by interoceptive hunger or satiety cues (Figure 3B).
Objective 3. Gating of competing needs by hunger circuits.
In a dynamic environment, organisms must balance the need to seek and consume food with other survival behaviors12. For example, an individual in energy deficit must prioritize seeking food over performing other protective behaviors. In the laboratory, we are modeling competing needs to explore how hunger circuits influence behavioral responses when an organism is faced with multiple conflicting signals. We recently discovered a link between hunger and pain, and this project will define how a novel hunger-related neural circuit gates the flow of noxious information from the spinal cord to the brain.
We reasoned that neural circuits coding for relatively acute survival needs, such as hunger, may influence less critical needs, like the long-term response to pain. As hunger builds, it stands to reason that an animal must ignore pain caused by a prior injury and venture out to seek food. In our studies, we demonstrated that while hunger has no impact on the response to acute pain (such as a hot surface), hunger reduces longer-term neuropathic or inflammatory pain responses (Figure 4A). This effect is mediated by a small subpopulation of AgRP neurons (~300 neurons)13 that project to the hindbrain parabrachial nucleus (PBN) to potently and selectively inhibit longer term pain associated with inflammation (Figure 4B). Further, we discovered that the inhibition of pain is mediated by the release of Neuropeptide Y (NPY) from the hypothalamic AgRP neurons, demonstrating that peptidergic signaling is acting on PBN neurons to filter nociceptive signals. Together, these findings suggest that hunger signals (via NPY signaling) converge with nociceptive information on a population of PBN neurons that are activated by pain14.

Our current experiments take a systematic approach to mapping, monitoring and manipulating the function of a proposed ‘three-neuron circuit’ that gates pain (Figure 4C). We are determining the contribution of specific PBN neuron populations -notably, with a focus on the neurons that express the receptor for NPY (NPY Y1R neurons). Ultimately, we will determine whether PBN NPY Y1R neurons are the site of convergence for hunger and nociceptive signals and will explore the circuit, cellular and molecular mechanisms that influence the perception and response of pain. These experiments will evaluate the function and physiology of an endogenous analgesic circuit that, if understood at a mechanistic level, could lead to novel pain management strategies.
References:
- Peacock, W. L. & Speakman, J. R. Effect of high-fat diet on body mass and energy balance in the bank vole. Physiology & behavior 74, 65-70, doi:10.1016/s0031-9384(01)00533-9 (2001).
- Speakman, J. R. A nonadaptive scenario explaining the genetic predisposition to obesity: the “predation release” hypothesis. Cell metabolism 6, 5-12, doi:10.1016/j.cmet.2007.06.004 (2007).
- Hetheringon, A. W., and Ranson, S.W. The relation of various hypothalamic lesions to adiposity in the rat. J. Comp. Neurol. 76, 475-499 (1942).
- Aponte, Y., Atasoy, D. & Sternson, S. M. AGRP neurons are sufficient to orchestrate feeding behavior rapidly and without training. Nature neuroscience 14, 351-355, doi:10.1038/nn.2739 (2011).
- Krashes, M. J., Koda, S., Ye, C., Rogan, S. C., Adams, A. C., Cusher, D. S., Maratos-Flier, E., Roth, B. L. & Lowell, B. B. Rapid, reversible activation of AgRP neurons drives feeding behavior in mice. The Journal of clinical investigation 121, 1424-1428, doi:10.1172/JCI46229 (2011).
- Luquet, S., Perez, F. A., Hnasko, T. S. & Palmiter, R. D. NPY/AgRP neurons are essential for feeding in adult mice but can be ablated in neonates. Science 310, 683-685, doi:10.1126/science.1115524 (2005).
- Su, Z., Alhadeff, A. L. & Betley, J. N. Nutritive, Post-ingestive Signals Are the Primary Regulators of AgRP Neuron Activity. Cell reports 21, 2724-2736, doi:10.1016/j.celrep.2017.11.036 (2017).
- Alhadeff, A. L., Goldstein, N., Park, O., Klima, M. L., Vargas, A. & Betley, J. N. Natural and Drug Rewards Engage Distinct Pathways that Converge on Coordinated Hypothalamic and Reward Circuits. Neuron 103, 891-908 e896, doi:10.1016/j.neuron.2019.05.050 (2019).
- Goldstein, N., McKnight, A. D., Carty, J. R. E., Arnold, M., Betley, J. N. & Alhadeff, A. L. Hypothalamic detection of macronutrients via multiple gut-brain pathways. Cell metabolism, doi:10.1016/j.cmet.2020.12.018 (2021).
- He, Z., Gao, Y., Alhadeff, A. L., Castorena, C. M., Huang, Y., Lieu, L., Afrin, S., Sun, J., Betley, J. N., Guo, H. & Williams, K. W. Cellular and synaptic reorganization of arcuate NPY/AgRP and POMC neurons after exercise. Molecular metabolism 18, 107-119, doi:10.1016/j.molmet.2018.08.011 (2018).
- Lieu, L., Chau, D., Afrin, S., Dong, Y., Alhadeff, A. L., Betley, J. N. & Williams, K. W. Effects of metabolic state on the regulation of melanocortin circuits. Physiology & behavior 224, 113039, doi:10.1016/j.physbeh.2020.113039 (2020).
- Tinbergen, N. The study of instinct. (Clarendon Press, 1951).
- Betley, J. N., Cao, Z. F., Ritola, K. D. & Sternson, S. M. Parallel, redundant circuit organization for homeostatic control of feeding behavior. Cell 155, 1337-1350, doi:10.1016/j.cell.2013.11.002 (2013).
- Alhadeff, A. L., Su, Z., Hernandez, E., Klima, M. L., Phillips, S. Z., Holland, R. A., Guo, C., Hantman, A. W., De Jonghe, B. C. & Betley, J. N. A Neural Circuit for the Suppression of Pain by a Competing Need State. Cell 173, 140-152 e115, doi:10.1016/j.cell.2018.02.057 (2018).
- Goldstein, N., McKnight, A. D., Carty, J. R. E., Arnold, M., Betley, J. N. & Alhadeff, A. L. Hypothalamic detection of macronutrients via multiple gut-brain pathways. Cell metabolism 33, 676-687 e675, doi:10.1016/j.cmet.2020.12.018 (2021).
- Chellappa, K., Brinkman, J. A., Mukherjee, S., Morrison, M., Alotaibi, M. I., Carbajal, K. A., Alhadeff, A. L., Perron, I. J., Yao, R., Purdy, C. S., DeFelice, D. M., Wakai, M. H., Tomasiewicz, J., Lin, A., Meyer, E., Peng, Y., Arriola Apelo, S. I., Puglielli, L., Betley, J. N., Paschos, G. K., Baur, J. A. & Lamming, D. W. Hypothalamic mTORC2 is essential for metabolic health and longevity. Aging Cell 18, e13014, doi:10.1111/acel.13014 (2019).
- Alhadeff, A. L., Park, O., Hernandez, E. & Betley, J. N. Inhibition of Itch by Hunger and AgRP Neuron Activity. Neuroscience 450, 126-134, doi:10.1016/j.neuroscience.2020.06.005 (2020).